
The pharmaceutical and life sciences industry is undergoing one of the most significant transformations in its history. Artificial intelligence is reshaping every stage of the drug development pipeline — from the initial identification of molecular targets through clinical trials, regulatory submission, and post-market surveillance — and its impact is extending beyond drug development into diagnostics, personalised medicine, and the way healthcare is delivered to patients. For an industry where the cost of bringing a single drug to market can exceed a billion dollars and the process can take more than a decade, the potential of AI to compress timelines, reduce costs, and improve success rates is genuinely transformative.
This article explores the key applications of AI across pharmaceuticals and life sciences, examines where the evidence for impact is strongest, and considers the challenges and responsibilities that come with deploying AI in one of the world’s most consequential industries.
AI in Drug Discovery: Compressing the Timeline From Target to Candidate
Drug discovery has traditionally been an extraordinarily slow and expensive process. Identifying a promising molecular target, screening millions of compounds for activity against that target, optimising lead compounds for potency and selectivity, and assessing their safety profile before they can even enter clinical testing — each stage involves enormous scientific complexity and significant attrition. The majority of compounds that enter drug discovery never reach patients. Those that do have survived a gauntlet of failures that have consumed years of research effort and vast resources.
AI is beginning to change this calculus fundamentally. Machine learning models trained on large databases of molecular structures, biological activity data, and clinical outcomes can predict with increasing accuracy how candidate compounds will behave — which are likely to bind effectively to their targets, which have favourable absorption and metabolism profiles, which are likely to be toxic. This predictive capability allows researchers to prioritise the most promising candidates earlier and deprioritise those likely to fail, reducing the volume of expensive laboratory work required and accelerating the path to viable drug candidates.

Generative Molecular Design
Perhaps the most exciting development in AI-assisted drug discovery is generative molecular design — the use of AI models to design entirely new molecules with specified properties, rather than simply screening existing compound libraries. Generative AI systems can explore vast regions of chemical space that no human medicinal chemist would think to investigate, proposing novel molecular structures optimised for potency, selectivity, and drug-like properties simultaneously.
Several AI-designed molecules have already entered clinical trials — a milestone that would have seemed implausible a decade ago. While it remains early days and the clinical success of AI-designed drugs will only be established over the coming years, the direction of travel is clear: AI is expanding the boundaries of what is chemically possible to pursue and accelerating the rate at which novel candidates can be identified and optimised.
Target Identification and Validation
Before molecules can be designed or screened, researchers must identify which biological targets are worth pursuing — which proteins, genes, or pathways, when modulated, are likely to produce therapeutic benefit without unacceptable side effects. This target identification and validation process is itself a major bottleneck in drug development, and AI is making significant contributions here too.
AI models can analyse genomic data, protein interaction networks, disease association databases, and scientific literature at a scale that far exceeds human capacity, identifying patterns that suggest novel target hypotheses. AlphaFold, DeepMind’s protein structure prediction system, has transformed structural biology by enabling accurate prediction of protein three-dimensional structures from sequence data alone — a capability that was previously accessible only through painstaking experimental work and that is now enabling drug discovery programmes that would previously not have been possible.
AI in Preclinical Development: Smarter Safety Assessment
Preclinical safety assessment — establishing that a candidate drug is safe enough to test in humans — is a critical and resource-intensive phase of drug development. Traditional approaches rely heavily on animal studies and in vitro laboratory tests, which are expensive, time-consuming, and imperfect predictors of human outcomes. AI is enabling new approaches to safety assessment that complement and in some cases may eventually reduce reliance on traditional methods.

Machine learning models trained on large toxicology datasets can predict the likelihood of specific safety signals — hepatotoxicity, cardiotoxicity, genotoxicity — from molecular structure alone, enabling safety assessment to begin at the earliest stages of drug design rather than only after candidate selection. Organoid models and organ-on-a-chip technologies, combined with AI analysis of the data they generate, are creating new in vitro systems that better represent human biology and provide richer safety data earlier in development.
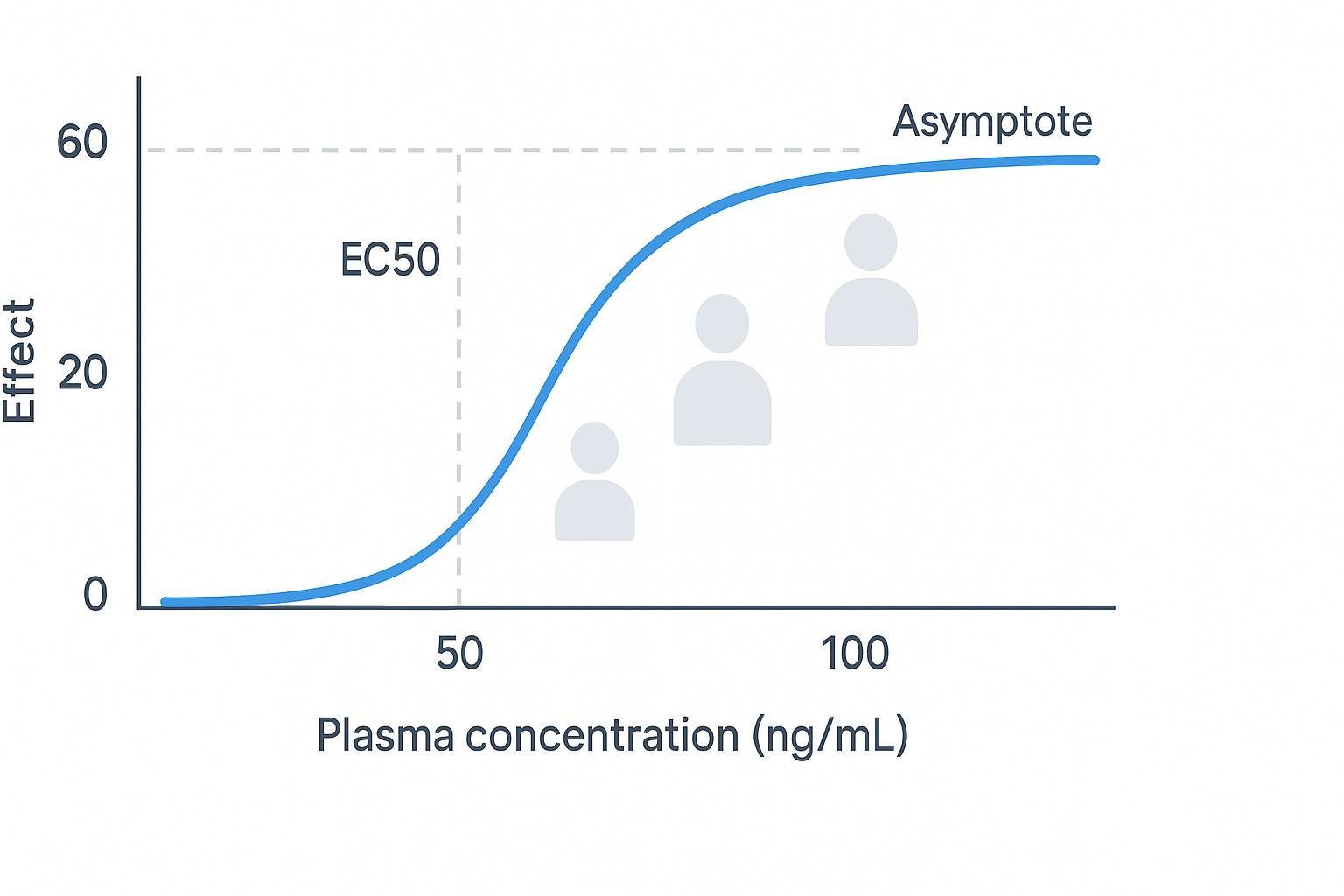
AI in Clinical Trials: Faster, Smarter, More Efficient
Clinical trials are the most expensive and time-consuming phase of drug development, and they are also the phase with the highest attrition rate — the majority of drugs that enter clinical testing fail to demonstrate sufficient efficacy or an acceptable safety profile to reach approval. AI is being applied across multiple dimensions of clinical trial design and execution to improve efficiency and increase the probability of success.
Trial Design and Protocol Optimisation
AI tools can analyse data from previous trials in related indications to inform decisions about optimal patient populations, dosing regimens, endpoints, and trial duration. Adaptive trial designs — where the protocol evolves based on emerging data during the trial itself — are becoming more practical as AI enables faster analysis of interim results. By designing trials more precisely from the outset and adapting them intelligently as data accumulates, AI is helping to reduce the sample sizes required and improve the likelihood of detecting genuine treatment effects.

Patient Recruitment and Retention
Finding and enrolling the right patients is one of the most significant operational challenges in clinical trials. AI systems can analyse electronic health records, genomic databases, and patient registries to identify potential trial participants who meet eligibility criteria — dramatically expanding the pool of candidates beyond what site-based recruitment alone can reach. AI tools can also monitor enrolled patients for signs of early dropout risk, enabling proactive outreach and support to improve retention rates and reduce the data loss that compromises trial integrity.

Real-World Evidence and Post-Market Surveillance
The regulatory approval of a drug is not the end of the evidence-generation process — it is the beginning of a new phase in which the drug’s performance in the real world, across a much broader and more diverse patient population than was studied in trials, must be monitored. AI is transforming pharmacovigilance and post-market surveillance by enabling continuous analysis of adverse event reports, electronic health records, social media, and other real-world data sources to detect safety signals that might not emerge in pre-approval studies.
AI in Oncology: Precision Medicine at the Frontier
Oncology is perhaps the area of medicine where AI is having its most dramatic near-term impact, and where the intersection of AI with pharmaceutical development is most apparent. Cancer is fundamentally a disease of molecular dysregulation, and the explosion of genomic, proteomic, and imaging data generated in oncology research creates exactly the kind of high-dimensional data environment where AI excels.
AI models are being used to identify biomarkers that predict which patients will respond to specific treatments, enabling oncologists to select therapies based on the molecular characteristics of each patient’s tumour rather than broad histological type. AI-powered analysis of pathology slides and medical imaging is enabling earlier and more accurate cancer detection. And AI is accelerating the development of entirely new classes of cancer therapies — including cell therapies and targeted agents — by enabling more sophisticated understanding of tumour biology and drug-target interactions.

Regulatory Dimensions: AI and the Approval Process
The integration of AI into pharmaceutical development raises important regulatory questions that agencies including the FDA, EMA, and MHRA are actively working to address. When AI is used to generate data or support decisions that feed into a regulatory submission, regulators need to understand how the AI system works, what its limitations are, how it was validated, and what human oversight was applied to its outputs. The opacity of many AI systems — particularly deep learning models — creates challenges for the explainability requirements that regulatory review depends on.
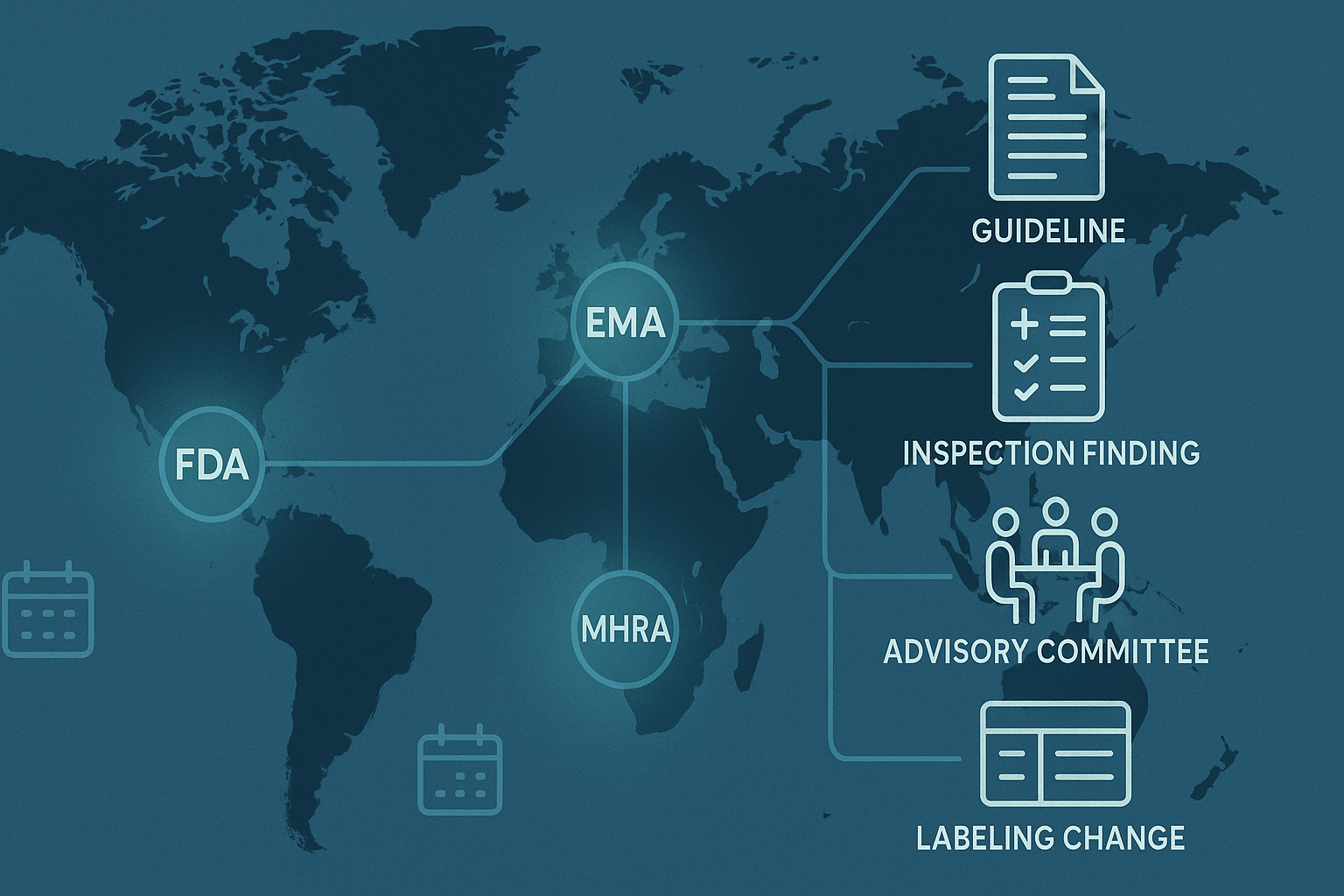
Regulatory agencies are developing guidance on the use of AI in drug development, and pharmaceutical companies are investing in the documentation, validation, and governance frameworks needed to support AI-assisted regulatory submissions. The direction is clear — AI will be embedded in the regulatory process as well as in development — but the frameworks for managing this transition responsibly are still being established.
The Patient at the Centre: AI and Personalised Medicine
Ultimately, the promise of AI in pharmaceuticals and life sciences is not about faster or cheaper drug development as an end in itself — it is about getting better treatments to patients more quickly, and ensuring those treatments are matched as precisely as possible to the individual patients most likely to benefit from them. The vision of personalised medicine — where treatment decisions are tailored to the molecular, genetic, and clinical profile of each individual — is becoming increasingly achievable as AI enables the analysis of the complex, multi-dimensional data that personalisation requires.

From companion diagnostics that identify the patients most likely to respond to a therapy, to AI-powered clinical decision support that helps oncologists select the optimal treatment sequence, to digital therapeutics that deliver personalised interventions through software — AI is enabling a vision of medicine that is more precise, more responsive, and more focused on the individual patient than was possible in the era of one-size-fits-all treatment.
For pharmaceutical professionals, life sciences researchers, clinical development teams, regulatory specialists, and healthcare leaders seeking comprehensive, authoritative coverage of how AI is reshaping every aspect of the pharmaceutical and life sciences sector — the AI Awareness guide to AI in pharmaceuticals and life sciences provides detailed, current, and practically oriented guidance on the full landscape of AI’s impact, from molecule to patient.
The Road Ahead: Responsible Innovation in a High-Stakes Industry
The potential of AI to transform pharmaceutical research and development is real and significant. But it comes with responsibilities that the industry must take seriously. The integrity of the evidence base for drug safety and efficacy must be maintained as AI becomes more deeply embedded in the processes that generate it. The benefits of AI-accelerated drug development must be accessible to patients across all geographies and economic circumstances, not only those in the wealthiest markets. And the human expertise — scientific, clinical, regulatory, ethical — that has always been at the heart of pharmaceutical development must remain central even as AI augments and accelerates the work.
The companies and research organisations that will lead the next era of pharmaceutical innovation are those that combine ambitious AI adoption with rigorous scientific standards, genuine patient focus, and the kind of responsible governance that ensures AI serves the ultimate purpose of medicine: improving and extending human life.